About Headaches
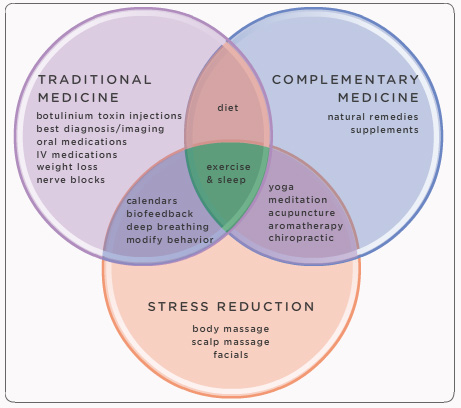 It is estimated that over 45 million people in the United States suffer from acute or chronic headaches,[1] with headaches accounting for one emergency room visit every 10 seconds. 18% of women and 8% of men between the ages of 18 and 55 suffer from what is technically defined as a migraine by the medical community. It is believed if those criteria are slightly relaxed, the number of individuals suffering from debilitating headaches is far greater. Headaches are the number two cause of disability in the United States, and headaches are one of the top causes for the loss of workplace productivity. Because the medical community has not yet fully embraced “headache” as a medical condition, it is believed that many headache sufferers remain undiagnosed.
It is estimated that over 45 million people in the United States suffer from acute or chronic headaches,[1] with headaches accounting for one emergency room visit every 10 seconds. 18% of women and 8% of men between the ages of 18 and 55 suffer from what is technically defined as a migraine by the medical community. It is believed if those criteria are slightly relaxed, the number of individuals suffering from debilitating headaches is far greater. Headaches are the number two cause of disability in the United States, and headaches are one of the top causes for the loss of workplace productivity. Because the medical community has not yet fully embraced “headache” as a medical condition, it is believed that many headache sufferers remain undiagnosed.
We care about our patients and their health. We continue to comply with all regulations to keep a clean and sanitized facility including required facemasks while in the office.
1. See migraineinformation.org for more information on this statistic.
This practice does not prescribe opioids, barbiturates, or controlled substances.
