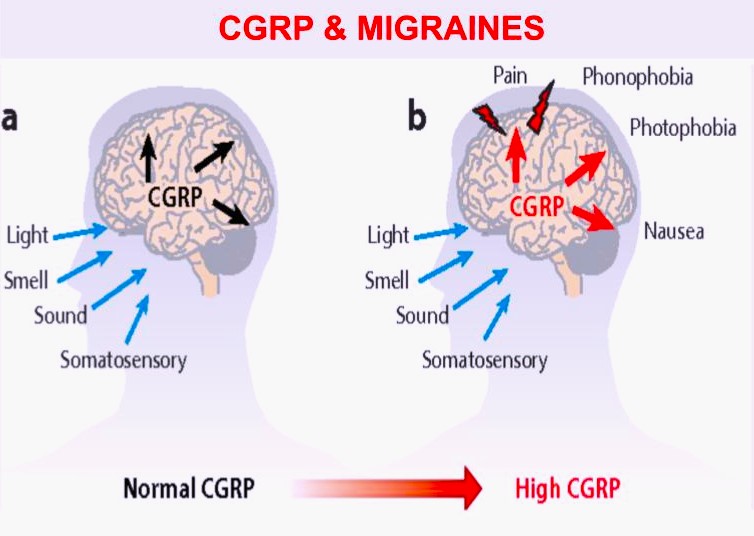
The calcitonin gene-related peptide (CGRP) is an amino acid neurotransmitter released by trigeminal ganglia nerves and is a potent vasodilator of cerebral vessels. CGRP plays a role in pain transmission from intracranial vessels to the central nervous system and affects vasodilation and neurogenic inflammation. Studies establish that CGRP release is part of the mechanism of migraine headaches. The FDA approved the monoclonal antibody CGRP antagonists erenumab, fremanezumab, and galcanezumab in 2018, eptinezumab in 2020, and rimegepant and atogepant in 2021 for migraine prevention. The FDA approved ubrogepant in 2019 and rimegepant in 2020 for treatment of acute migraine in adults. Please make an appointment with a MCHN provider to discuss whether these effective CGRP migraine preventative and abortive medications are appropriate for your pain.
–Alice Wong, NP
Reference: www.uptodate.com/pathophysiology-clinical-manifestations-and-diagnosis-of-migraine-in-adults
